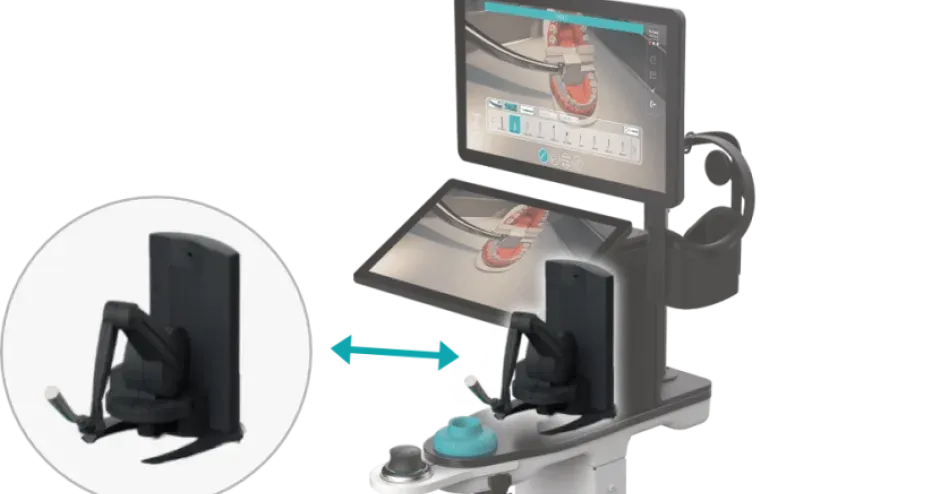
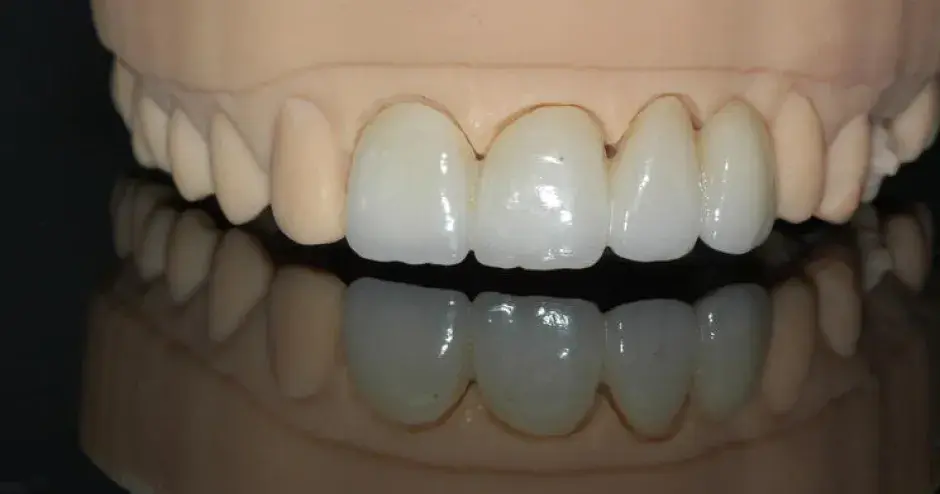
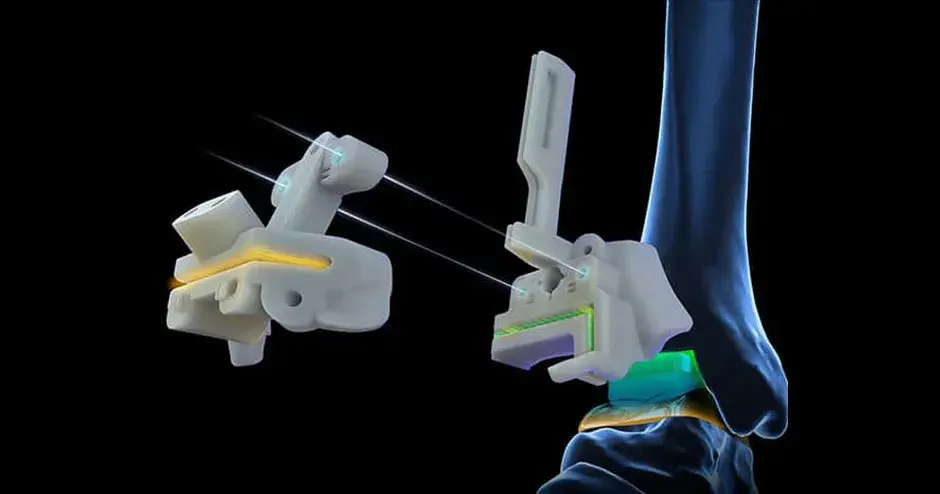
“We are learning a lot very quickly, particularly in the area of respiratory systems so that, whenever possible, we can translate urgent requests from hospitals into practical part solutions that can be used to support their life-saving efforts.”— Riccardo Nicastro, Business Development Manager Southern Europe – 3D Systems
Disclaimer
The production of medical parts requires conformance with certain regulations. The designs and parts discussed above do not have regulatory clearance at this time. Any required validations and regulatory approvals are not the responsibility of 3D Systems but are the responsibility of the customer and/or user of the parts. All parts ordered from 3D Systems are subject to 3D Systems’ Standard Terms and Conditions for Part Production Services available at https://www.3dsystems.com/part-production-services-terms-conditions.
Any design files or parts referenced or provided by 3D Systems relating to COVID-19 response support are provided “as-is” without any warranties or guarantees of any kind, including but not limited to, warranties of fitness for particular purposes, merchantability, or non-infringement. Design files for the manufacture of parts for medical use do not include any registrations or certifications. Users of finished parts or design files are solely responsible for their use and any applicable regulatory approvals (or exceptions thereto) and any claims of liability, including but not limited to products liability or intellectual property infringement, and will indemnify 3D Systems for any liability that may result from use of the design files or parts.