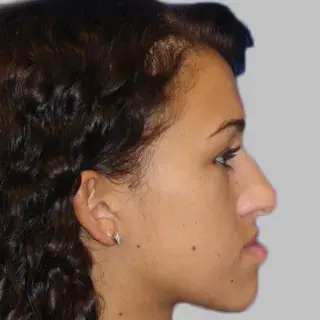
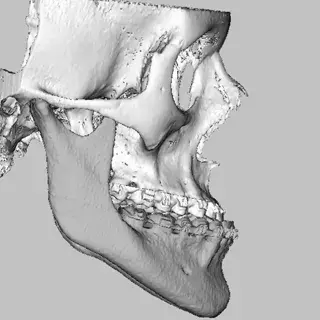
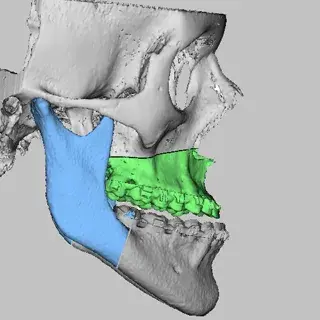
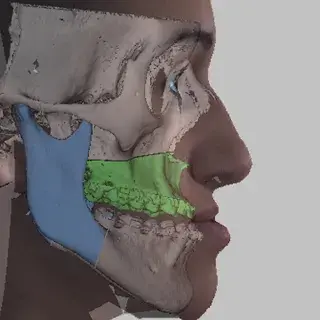
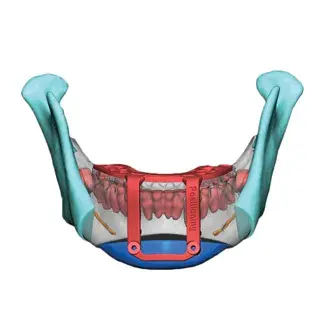
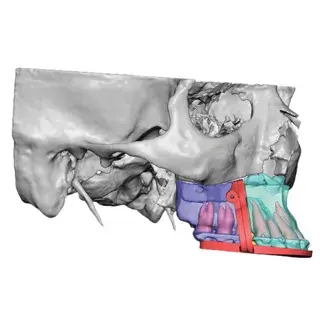
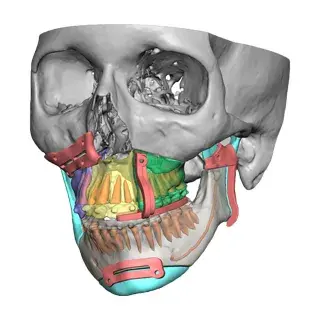
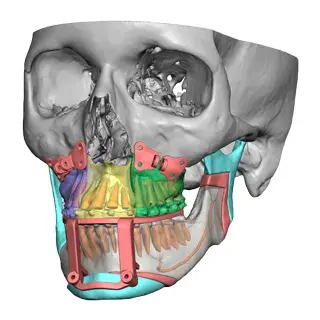
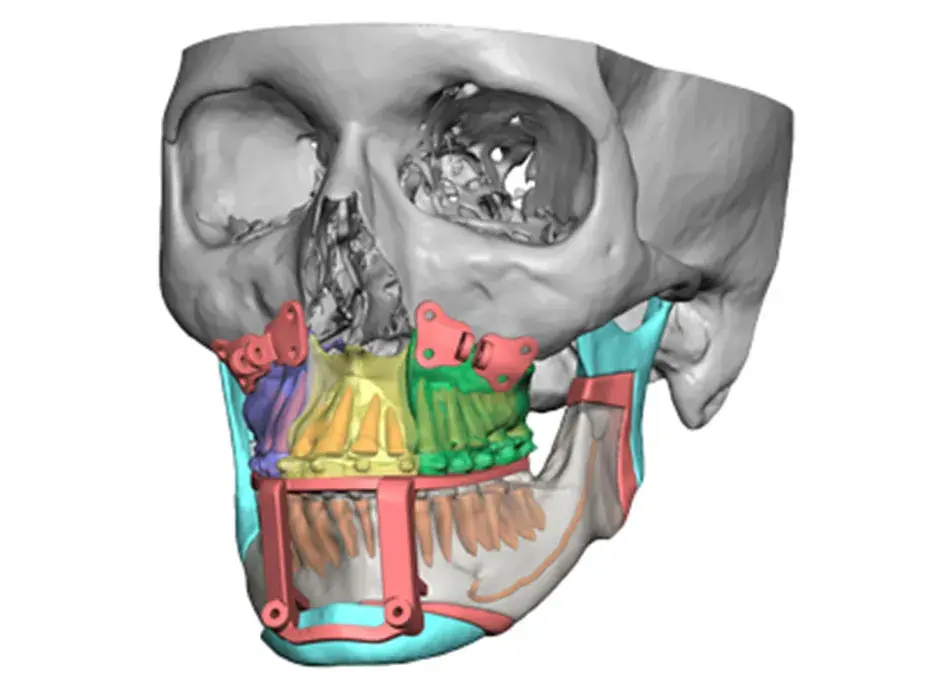
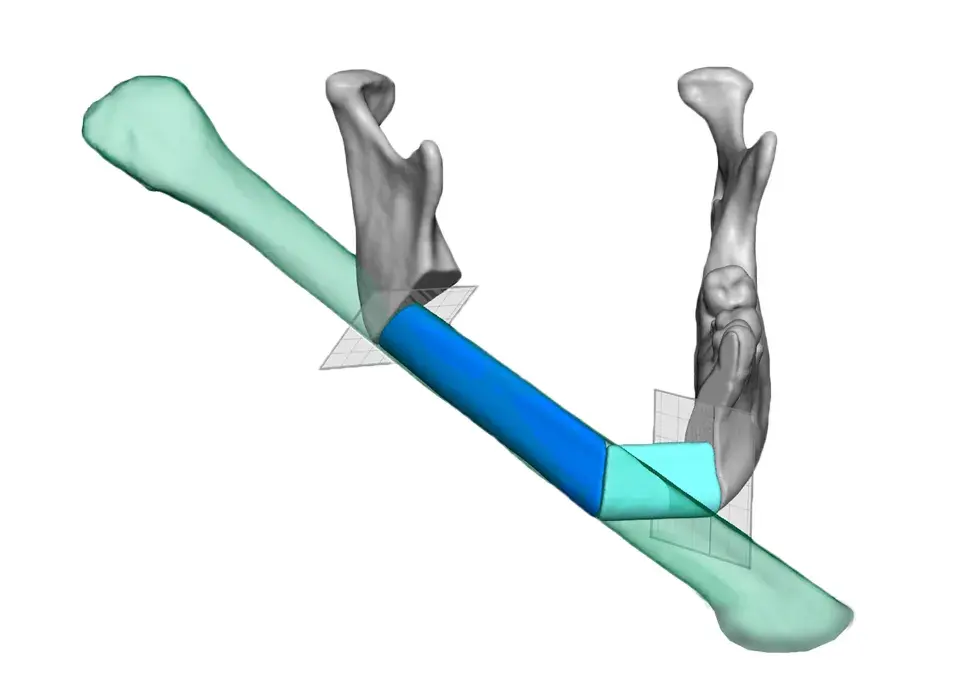
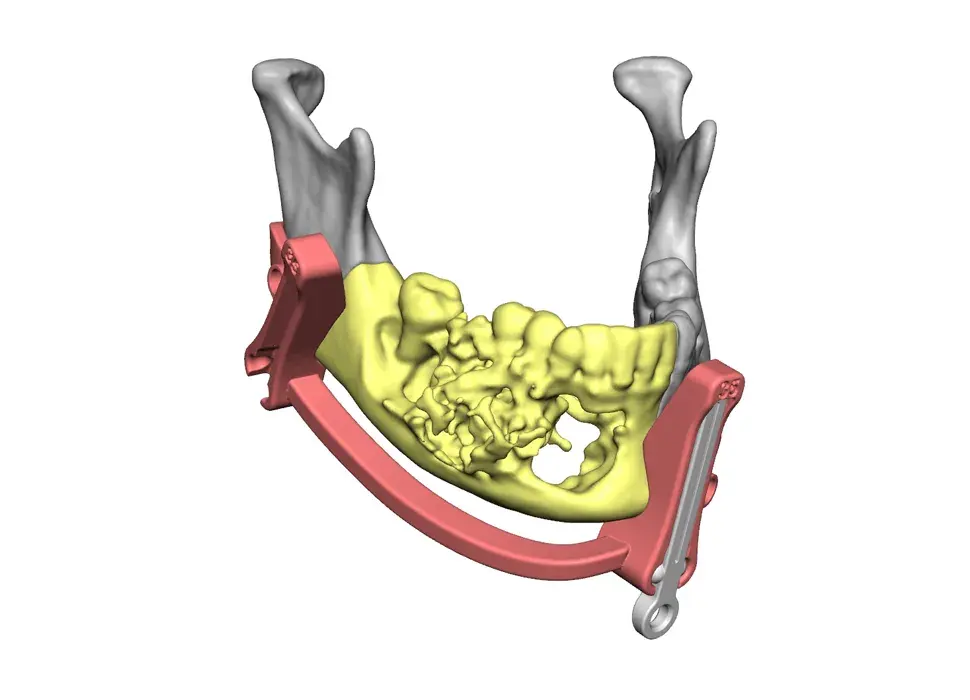
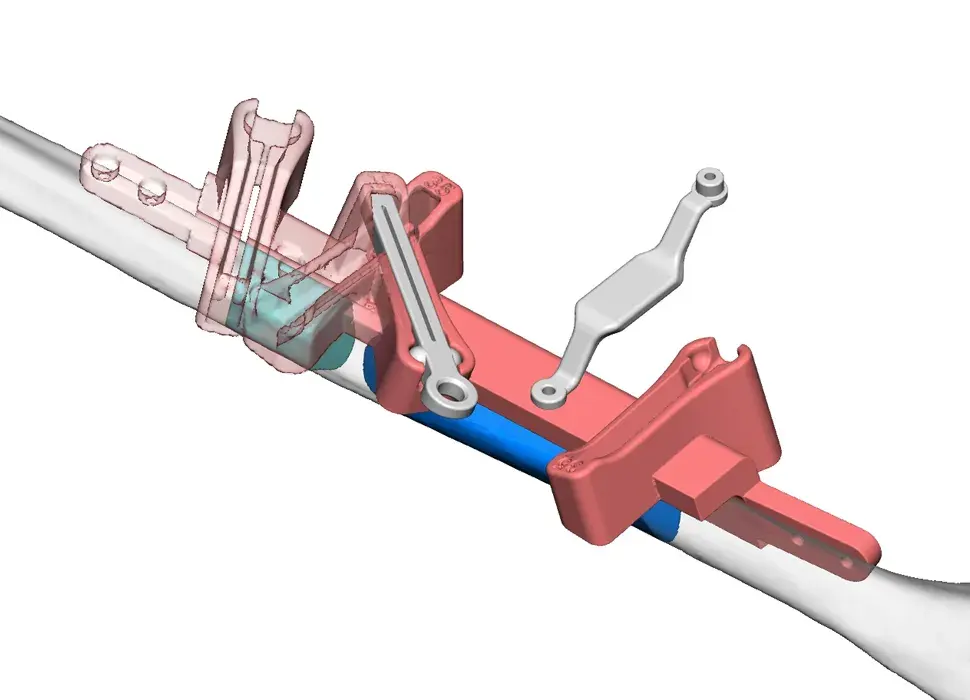
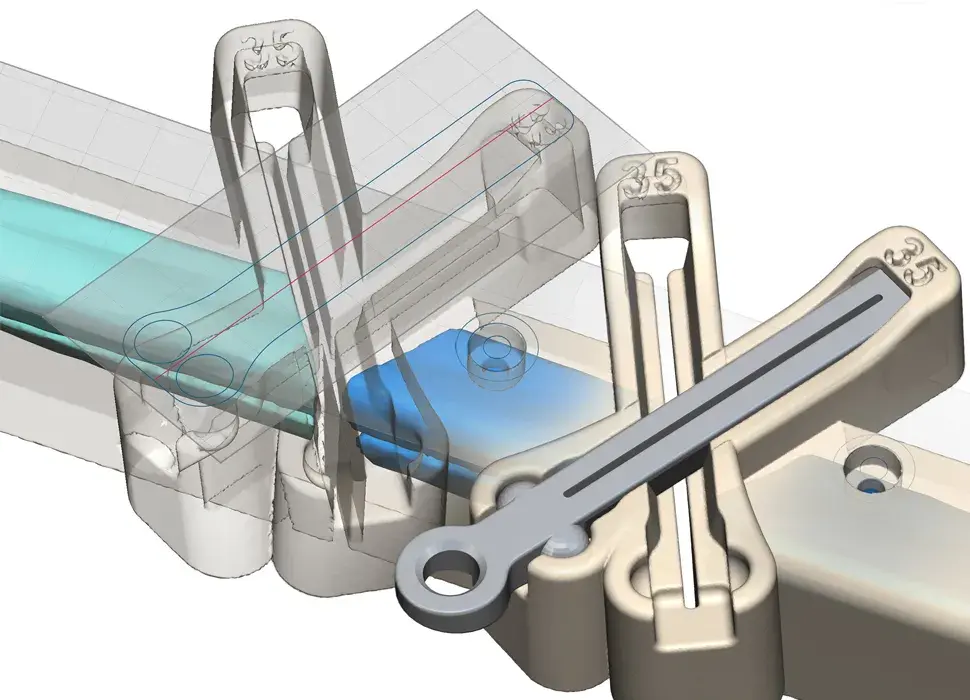
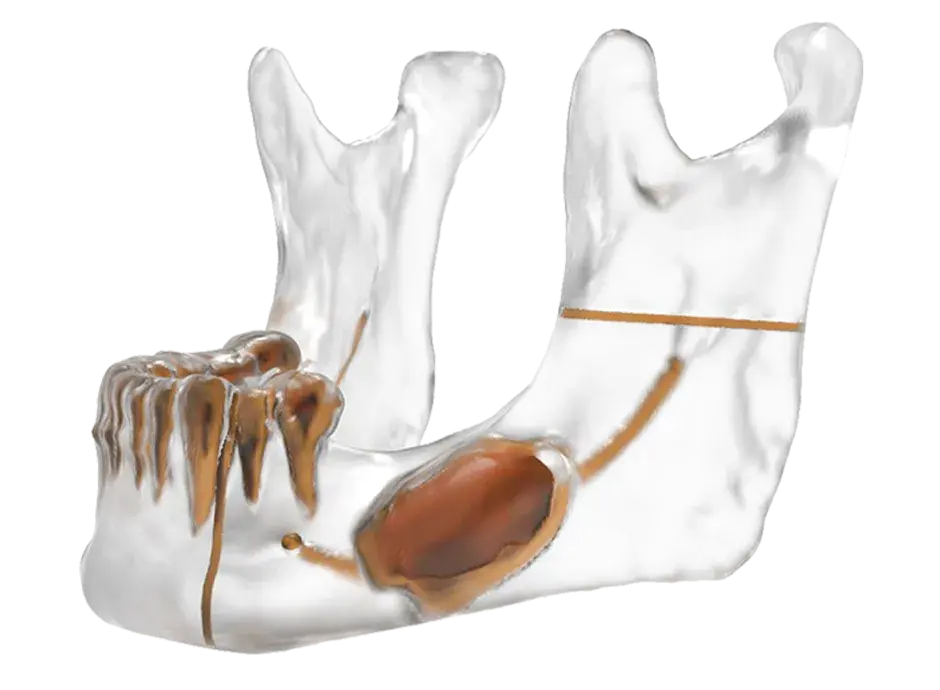
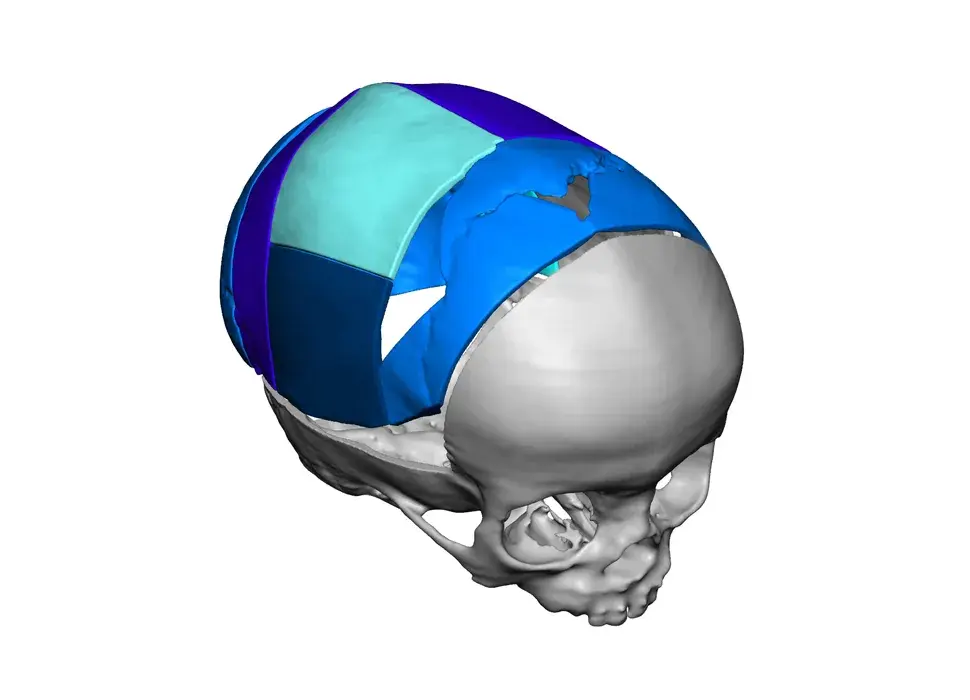
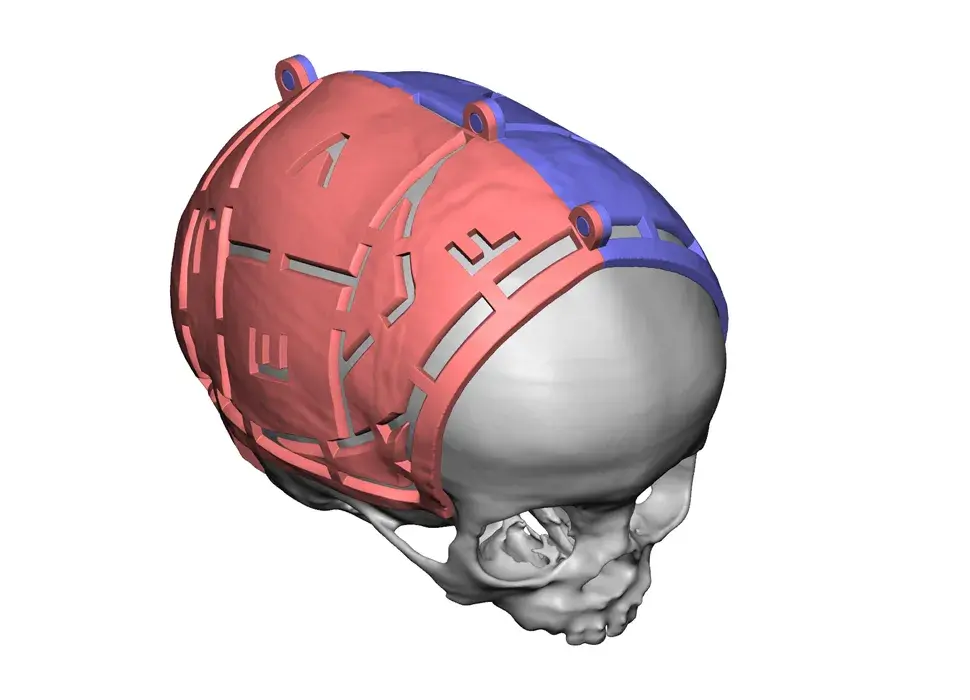
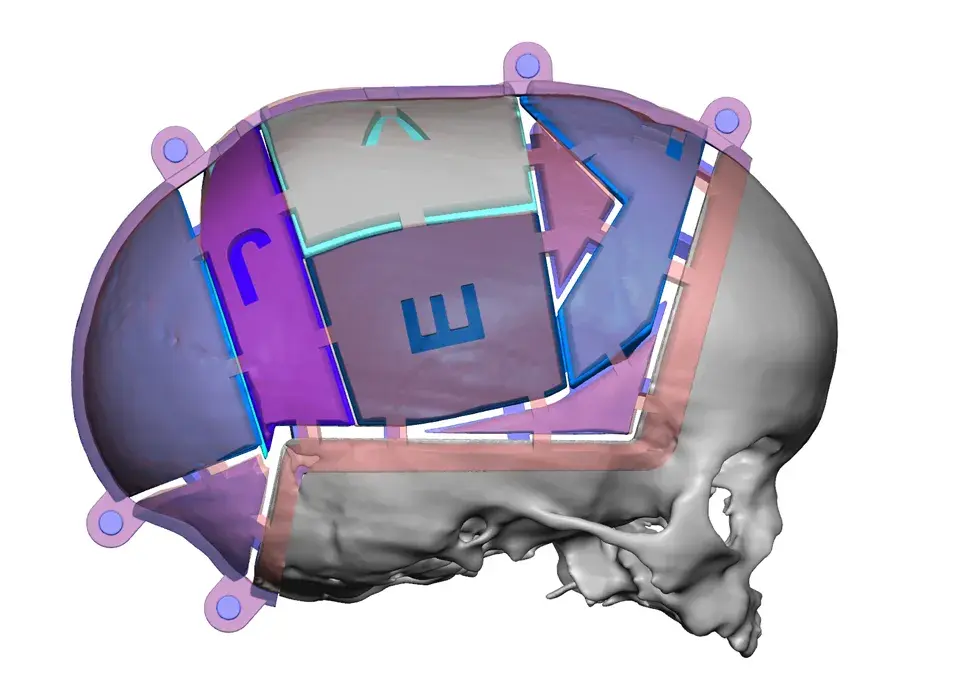
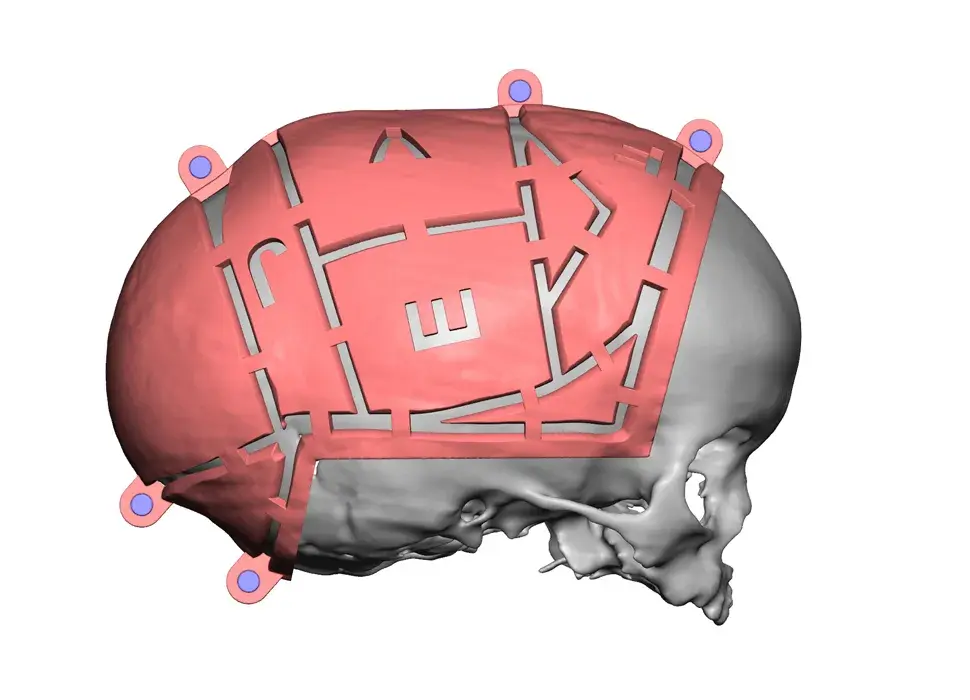
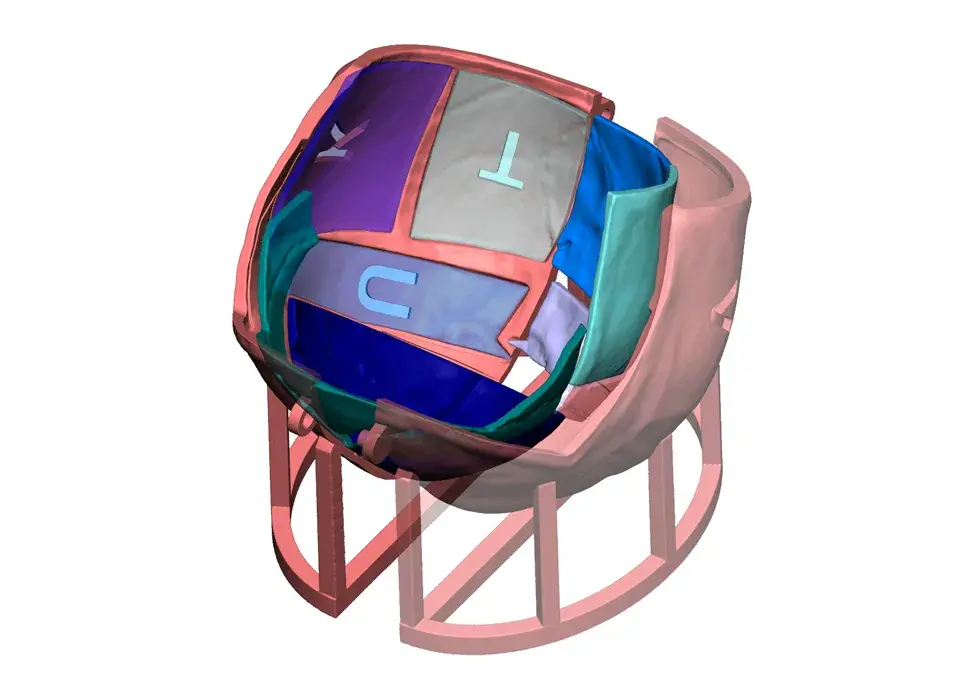
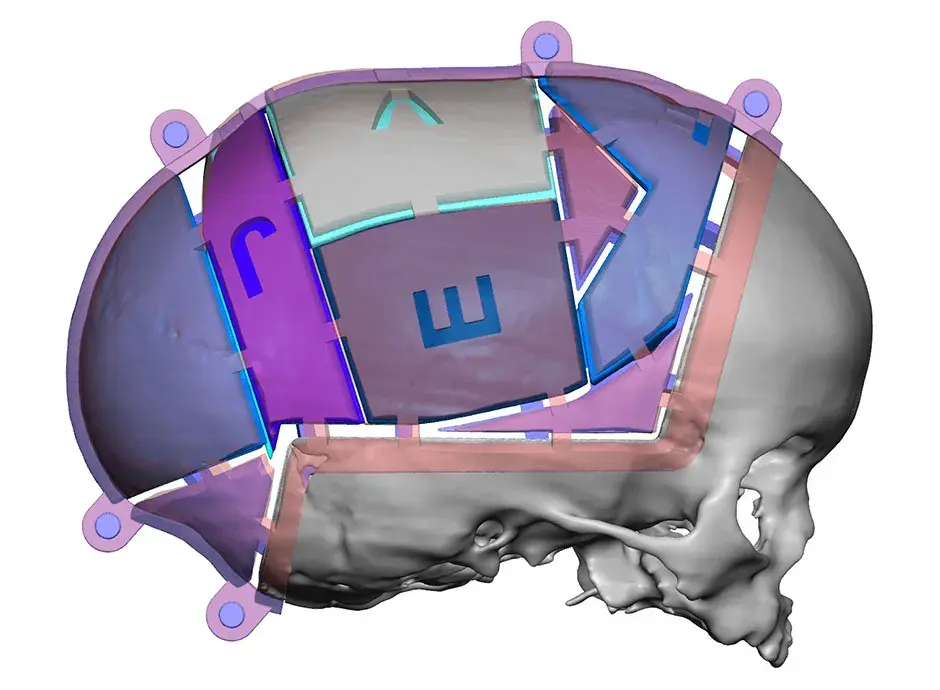
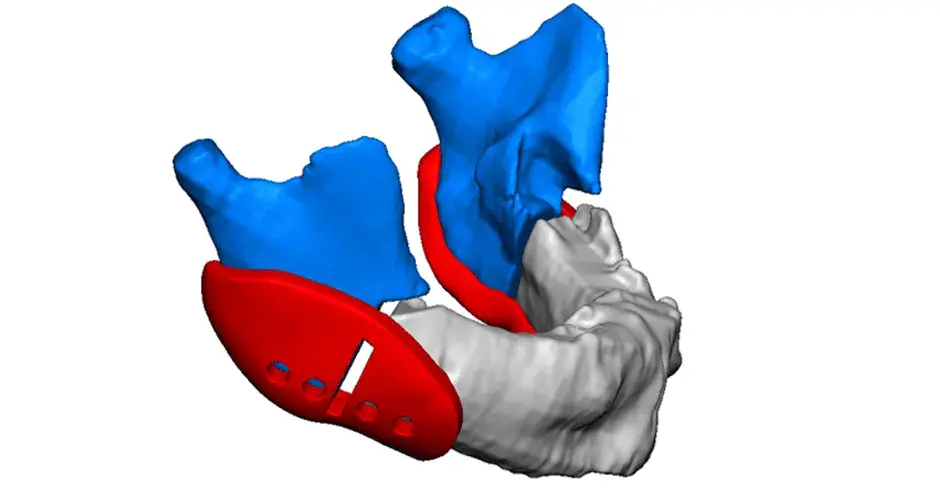
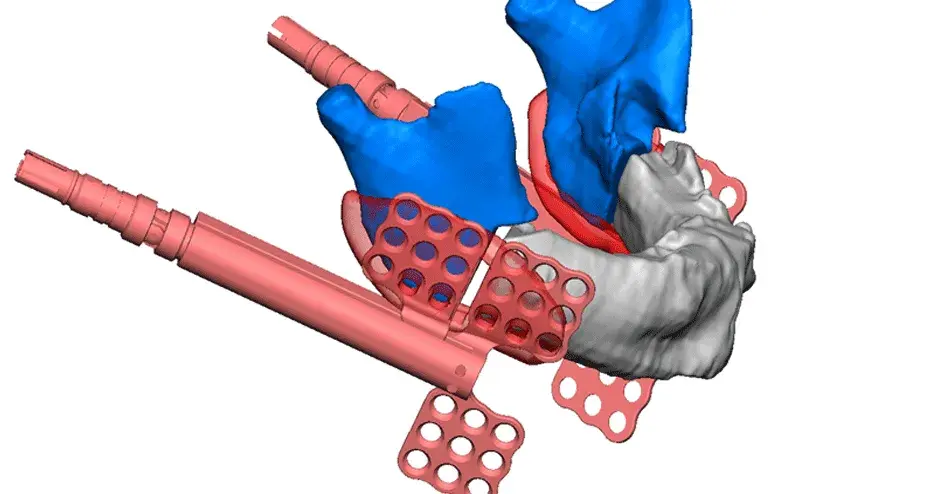
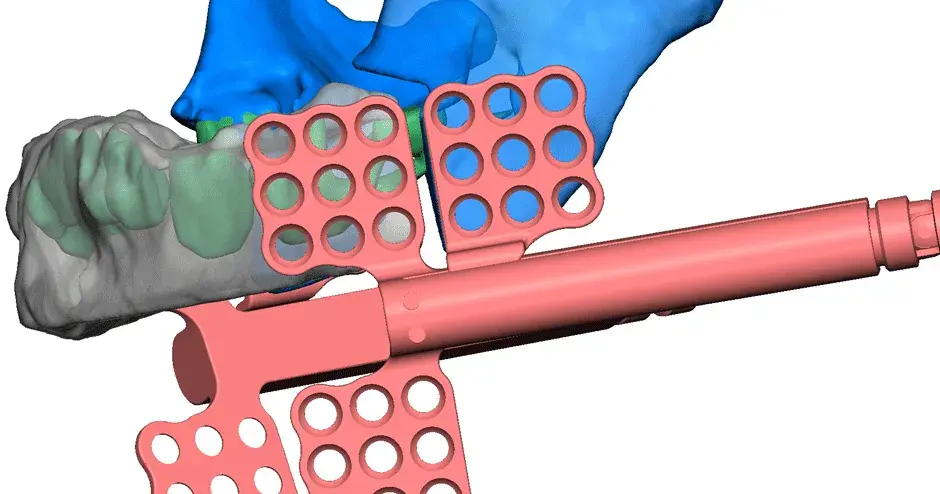
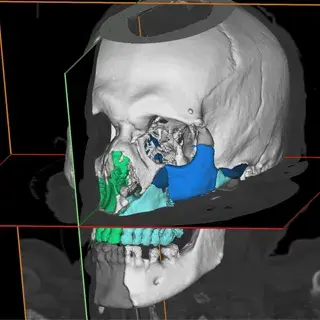
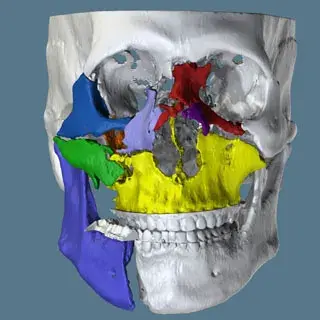
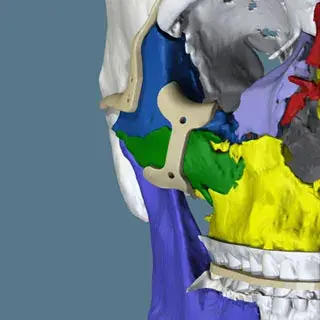
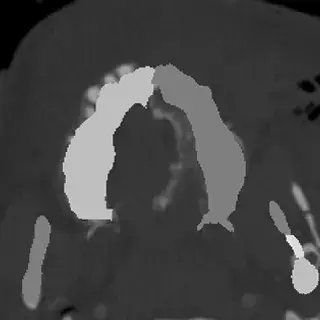
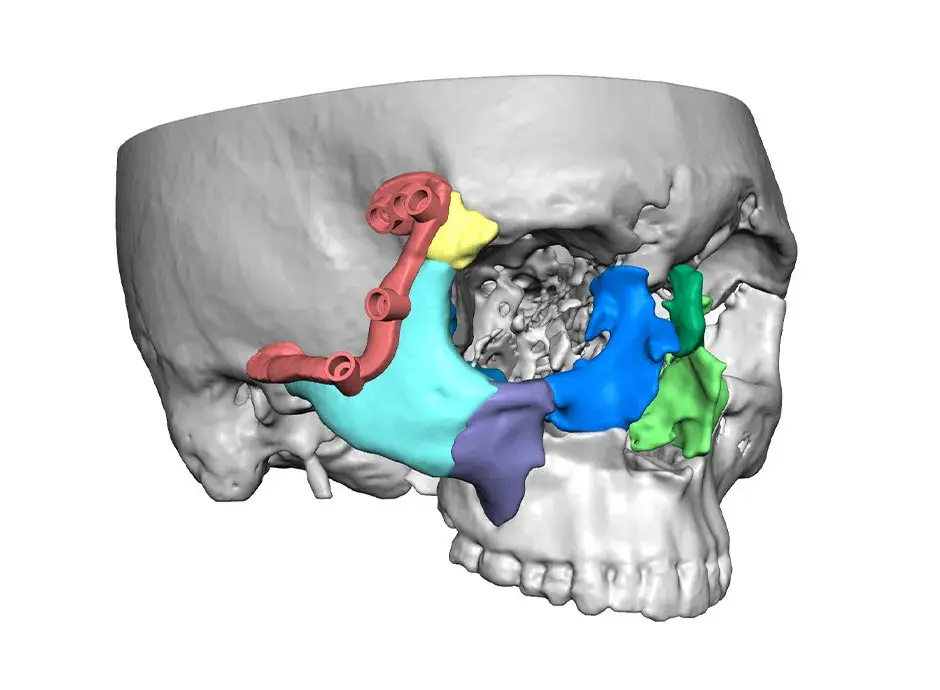
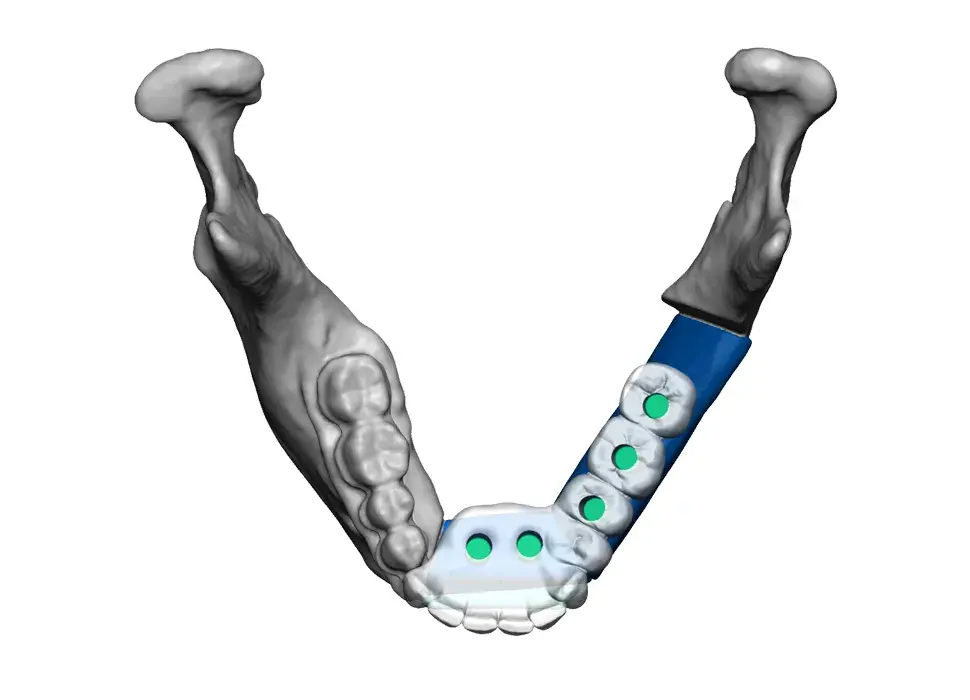
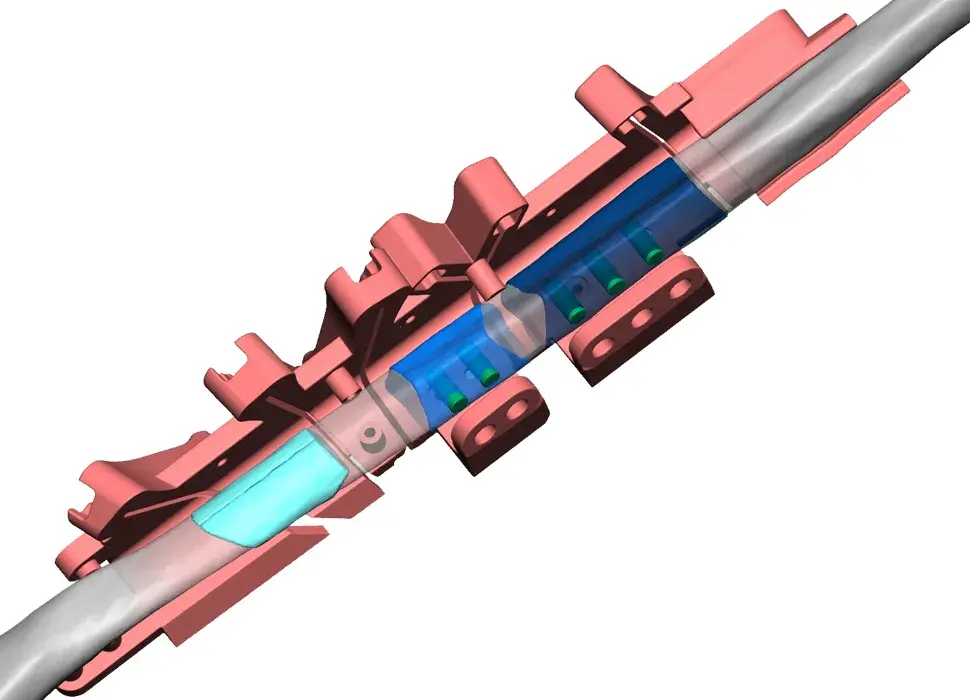
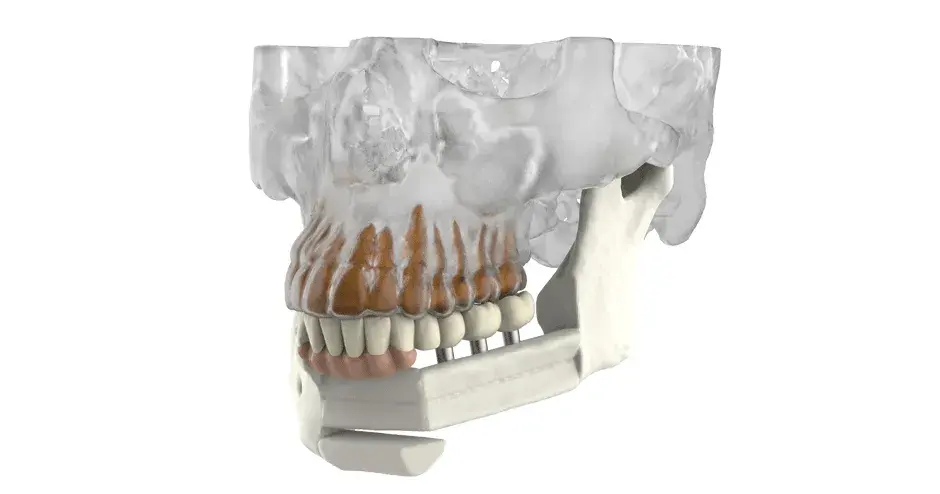
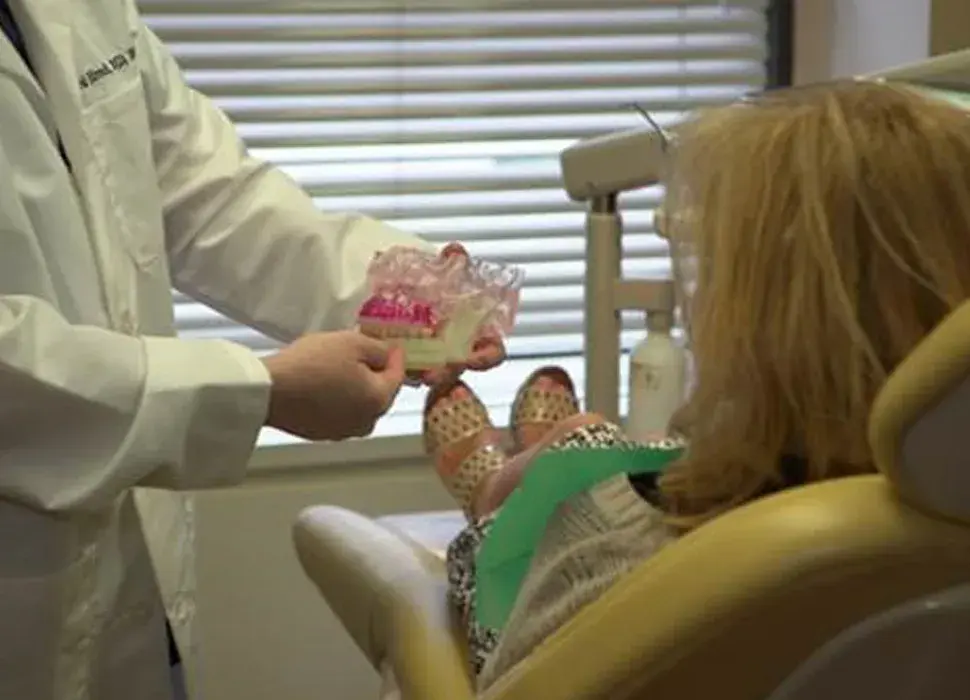
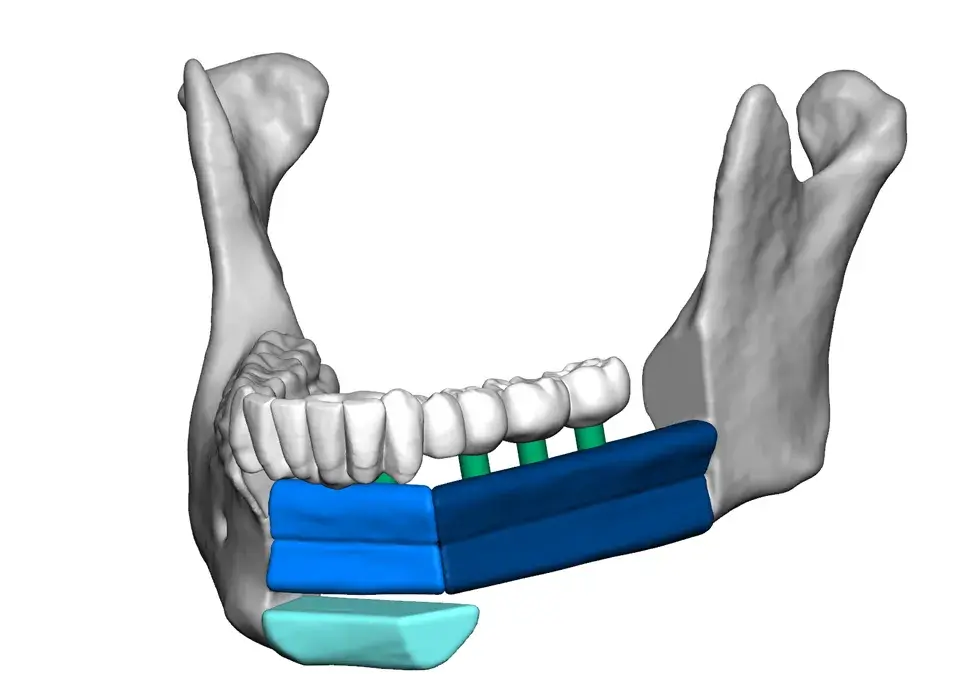
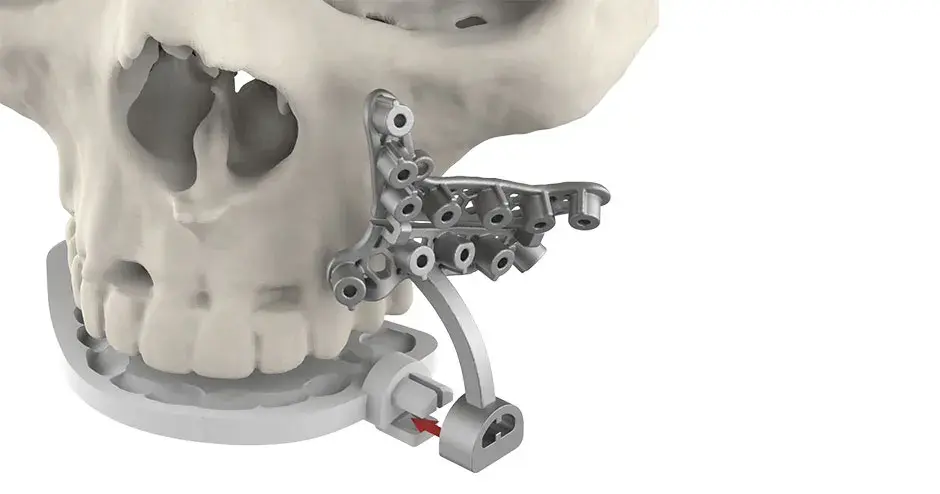
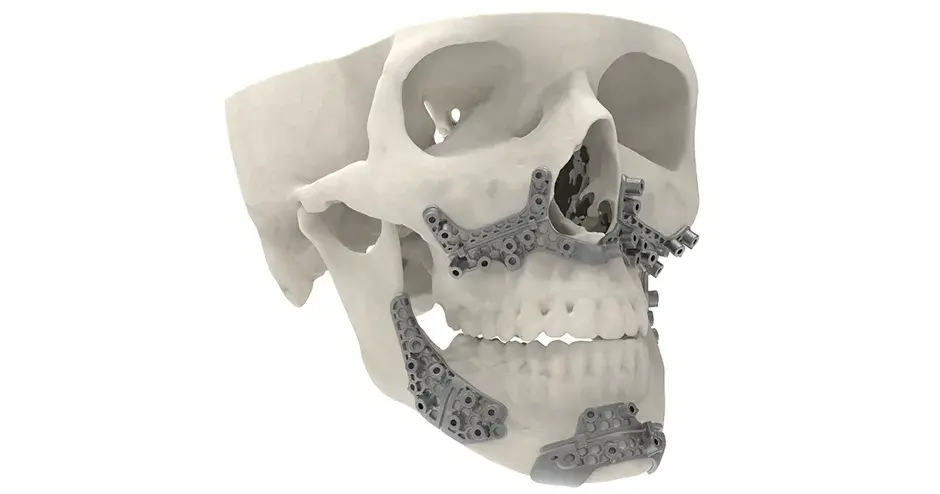
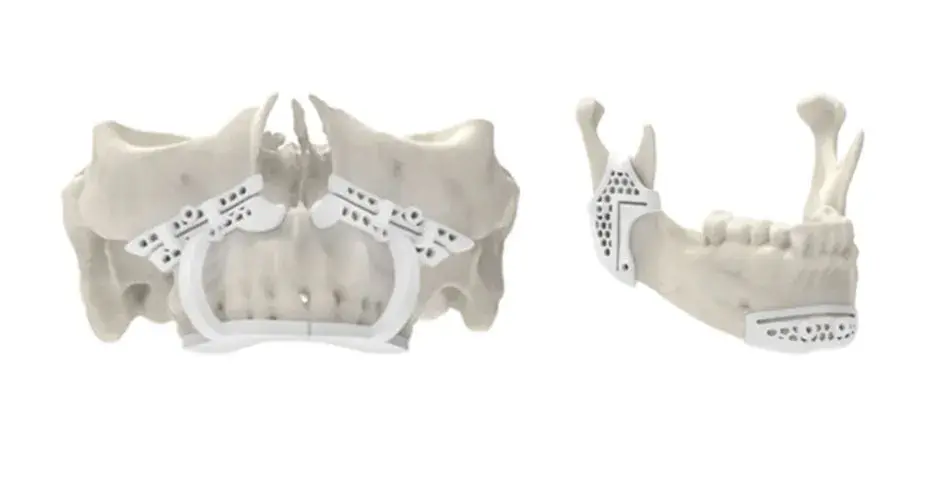
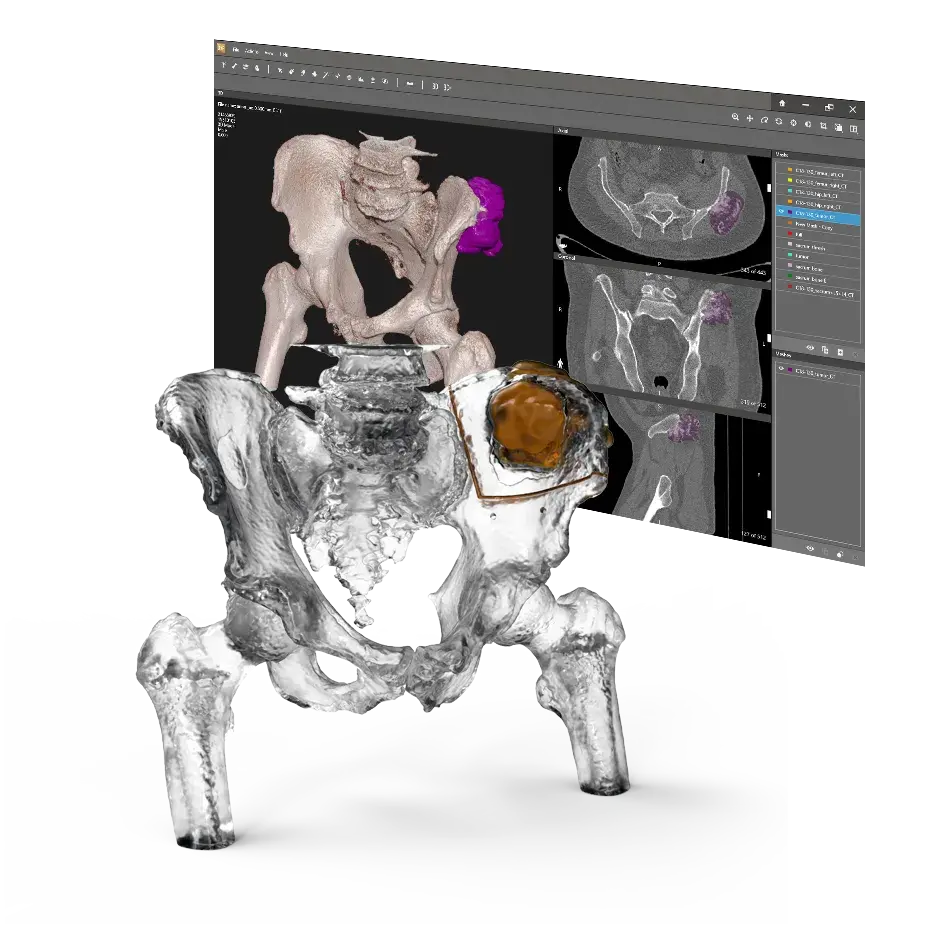
3D Systems' VSP® surgical planning solutions for craniomaxillofacial (CMF) applications received FDA clearance as a service-based approach to personalized surgery over 10 years ago.
3D Systems and Stryker Corporation have partnered to provide surgeons with best-in-class products and services for craniomaxillofacial surgeries. As a leader in personalized healthcare solutions, 3D Systems has planned and delivered devices for more than 140,000 patient-specific cases. The Stryker Craniomaxillofacial business specializes in providing patient-specific options and innovative solutions that help drive efficiencies in surgical suites. The combination of Stryker’s specialized team and advanced implants with 3D Systems' cutting-edge 3D printing technologies and expert consulting services positions both companies to provide a superior level of service to healthcare professionals who use these revolutionary solutions.