A Q&A with Gautam Gupta, vice president, business development and global sales, healthcare for 3D Systems.
What services are offered by 3D Systems’ Medical Device Design and Manufacturing Group?
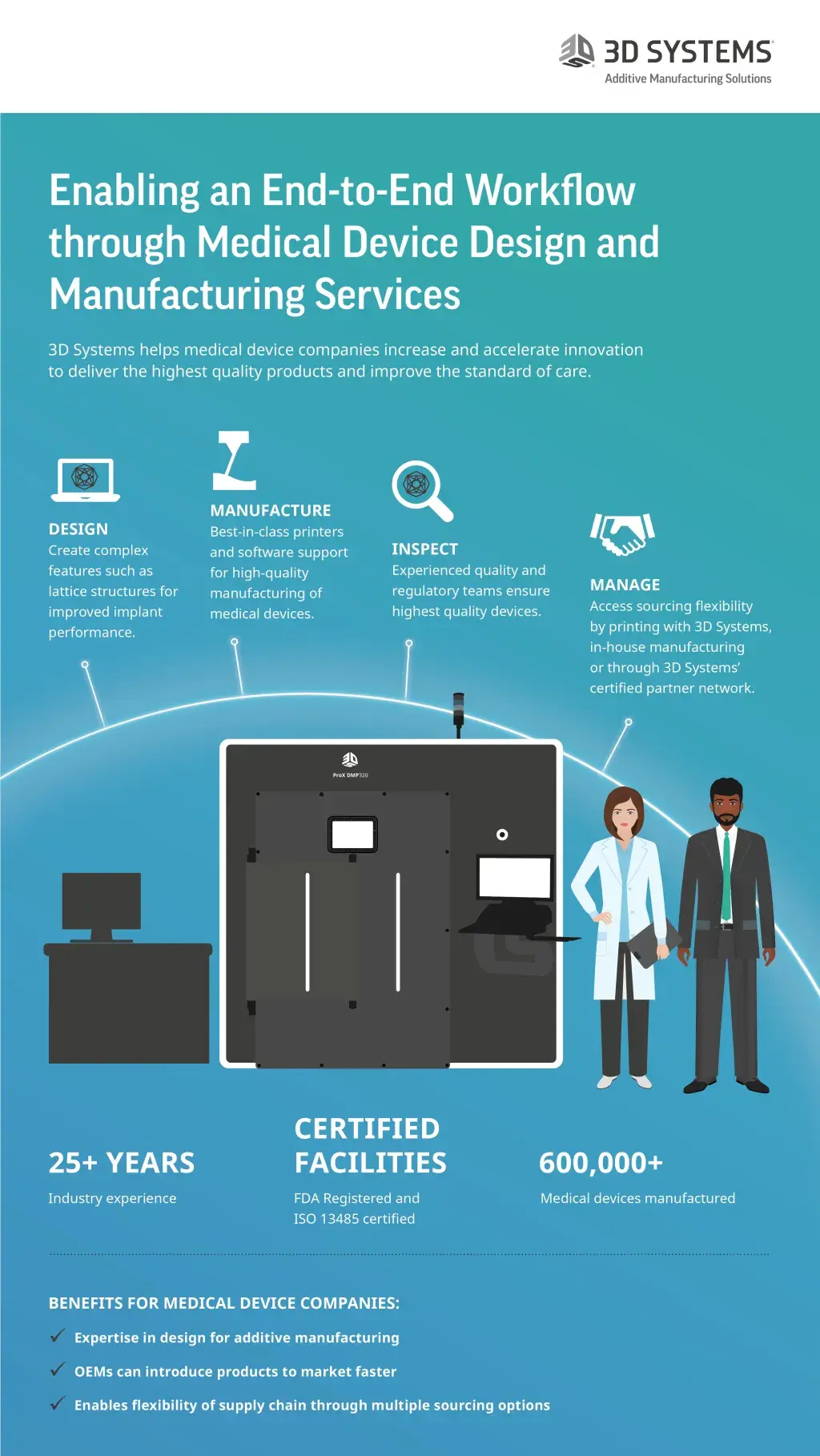
3D Systems’ medical device design and manufacturing service is truly a unique offering. With two ISO 13485 and FDA registered facilities strategically located in the US and Europe, we are able to support customers globally. Our team takes pride in not being just a contract manufacturer, instead, we work with medical device companies every step of the way from concept development to commercialization of the product.
The process begins with one-on-one interaction with our skilled applications engineers. These engineers have deep knowledge of the medical device industry as well as 3D printing. They collaborate with the OEMs to optimize the design of devices for 3D printing on our fleet of state-of-the-art direct metal printers. Our service doesn’t end there, we continue to support customers during their submission for FDA clearance of their devices and pride ourselves in the ability to support getting their products to market faster.
With the addition of our certified partner program, we now offer medical device companies unmatched flexibility with multiple strategic sourcing options. The OEMs can source their parts directly from us, or from our certified partner or do it in-house using the same printers on which their parts were developed and cleared in the first place.
What makes your group unique compared to competitors?
Our end-to-end solutions set us apart. We are uniquely positioned to partner with medical device companies addressing orthopedic and spinal needs due to our end-to-end metal additive solutions that include state-of-the-art metal 3D printing with the ProX DMP 320 printers, 3DXpert design for additive manufacturing software, and proven LaserForm® materials. Our experience in assisting with getting dozens of medical devices cleared through the regulatory process also provides companies a unique advantage and assurance to bring this revolutionary technology to the patient’s bedside in the operating room. In addition, through our certified partner program, OEMs have the benefit of sourcing flexibility and risk mitigation which no other company can match.
What trends are you seeing and what do those trends mean for the broader healthcare industry?
The medical device industry is starting to really understand the power of 3D printing in optimizing the design of implants and instruments. The industry is trying to leverage the power of additive software and hardware to reduce the number of processing steps and components in a device and thereby reduce the overall cost of manufacturing a device or an instrument.
These trends mean that the healthcare industry should be able to leverage the power of 3D printing technology to fuel innovation and bring revolutionary products to the market to improve patient care. We can see this revolution happening in the spine industry right now where the interbody fusion devices used to be almost exclusively made used materials like PEEK and manufactured using traditional subtractive manufacturing processes. But now the spine industry is adopting 3D printing to introduce new revolutionary products that can promote bone ingrowth and thereby improve implant fixation to host bone. The printing of these devices also reduces the number of manufacturing steps, thereby making the additive process more cost effective in several cases. The idea of fueling innovation to bring highly effective products at reduced overall cost that can improve the standard of care is revolutionary.
What are medical device OEMs asking for most often when it comes to 3D printing for their orthopedic-related products?
Since 3D printing technology has only recently been adopted by the industry for large-scale manufacturing, there is still a learning curve when it comes to using this technology. The medical device industry most critically needs an experienced partner that can help them optimize their workflow to enable the introduction of high quality, innovative products to the market in a rapid, reliable and cost-effective way.
At 3D Systems, we have two world-class ISO 13485-certified manufacturing centers located strategically in Denver, Colorado and Leuven, Belgium to support the customers in the US and Europe. Our expert teams are supporting large-scale production of orthopedic and spine devices from these centers using our state-of-the-art solutions. This success has allowed us to gain trust and credibility with our partners in the medical device industry.

Any challenges with new or advanced materials? How rapidly are these evolving?
Qualifying new materials for 3D printing technology in healthcare presents challenges at multiple levels. First, the material has to be qualified for its biocompatibility and ability to meet mechanical requirements for multiple applications. The regulatory and financial burden of such qualification and the associated time can be substantial. Although still a challenging task from a logistics and technology standpoint, it is faster, easier and less-expensive to qualify already proven materials like 17-4 PH stainless steel and validate those for new printing applications.
The second challenge is in regards to machine hardware engineering. Validating and optimizing parameters to run new materials can be challenging and costly unless there is a business case for a clinical application that requires large-scale manufacturing.
Third, securing a logistics ecosystem for reliable sourcing of such new materials at the quality required for healthcare applications can also be a challenging task.
3D Systems is well positioned to help medical device manufacturers overcome these challenges. Our expertise combined with a diverse, broad, library of biocompatible materials enable our healthcare teams to assist in more easily streamlining these processes for partners and customers.
Want to find out more?
Want to learn more about our Medical Device Design & Manufacturing services? Click on the infographic at the top of the page to explore our end-to-end workflow, or visit https://www.3dsystems.com/medical-device-manufacturing